Phenylsulfanyltetrafluorobromoethane
| Cat. # | Quantity | Price | Lead time | Buy this product |
|---|---|---|---|---|
| FBR001_1 | 1 g | 450,00 € | in stock | |
| FBR001_5 | 5 g | 1.250,00 € | in stock | |
| FBR001_10 | 10 g | 1.800,00 € | in stock |
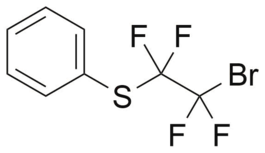
- IUPAC Name: (2-Bromo-1,1,2,2-tetrafluoroethyl)(phenyl)sulfane
- Synonyms: PhSCF2CF2Br
- CAS: 83015-33-2
- Smiles: FC(F)(C(F)(Br)F)SC1=CC=CC=C1
- Chemical formula: C8H5BrF4S
- Molecular weight: 289.09
- Purity: 95%+
A phenylsulfanyltetrafluoroethyl moiety incorporated in the substrate can be treated with tributyltin hydride and generate the corresponding fluoroalkyl radical. If the substrate lacks any olefine, it can be reduced to tetrafluoroethyl group. If the substrate contains an olefine in the correct spatial orientation, this can lead to an intramolecular cyclisation affording tetrafluorinated cyclic structures.
