Pentafluoroethyl azide 0.15 M solution in THF
| Cat. # | Quantity | Price | Lead time | Buy this product |
|---|---|---|---|---|
| FAZ002_5 | 5 ml | 335,00 € | in stock | |
| FAZ002_10 | 10 ml | 490,00 € | in stock | |
| FAZ002_25 | 25 ml | 990,00 € | in stock |
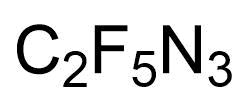
- IUPAC Name: Azidopentafluoroethane
- Synonyms: 1-Azido-1,1,2,2,2-pentafluoro-ethane, C2F5N3, Pentafluoroethyl azide, Perfluoroethyl azide
- CAS: 2055167-74-1
- Smiles: FC(F)(N=[N+]=[N-])C(F)(F)F
- Chemical formula: C2F5N3
- Molecular weight: 161.034
Recently, a new, elegant and scalable synthesis of pentafluoroethylazide (C2F5N3) solution in THF has been developed in Dr. Beier group at IOCB Prague in collaboration with CF Plus Chemicals (EP2982672A1).1 These otherwise difficult-to-access perfluoroalkylazides are highly reactive in copper catalyzed alkyne-azide cycloaddition reaction affording stable N-C2F5 triazoles in a highly regioselective manner.
The efficiency of formation of these virtually unexplored perfluoroalkyltriazoles make them attractive for drug and pesticide discovery programmes. This chemistry was highlighted in Chemical and Engineering News in December 2016. Later, our collaborators demonstrated that the triazoles prepared by copper catalyzed azide-alkyne cycloaddition can be transformed into a plethora of hitherto unreported five-membered N-(per)fluoroalkyl heterocycles.2
Densely functionalized 4,5-disubstituted N-perfluoroalkyltriazoles could be also accessed by enamine-mediate azide-ketone [3+2] cycloaddition using easily enolisable ketones in the presence of catalytic amounts of pyrrolidine.3
References:
1) Angew. Chem. Int. Ed., 2017, 56, 346.
2) Chem. Commun., 2018, 54, 3258-3261.
3) Chemistry Select, 2018, 3, 7045-7048.
