Ethyl difluoroazidoacetate
| Cat. # | Quantity | Price | Lead time | Buy this product |
|---|---|---|---|---|
| FAZ008_0050 | 0.05 g | 100,00 € | in stock | |
| FAZ008_0100 | 0.1 g | 150,00 € | in stock | |
| FAZ008_0250 | 0.25 g | 250,00 € | in stock |
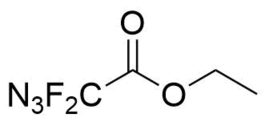
- IUPAC Name: Ethyl 2-azido-2,2-difluoroacetate
- Synonyms: Ethyl 2,2-difluoro-2-azidoacetate, Ethyl 2-azido-2,2-difluoroacetate, Ethyl difluoroazidoacetate, 2-azido-2,2-difluoroacetic acid ethyl ester
- CAS: 153755-61-4
- Smiles: O=C(C(F)(N=[N+]=[N-])F)OCC
- Chemical formula: C4H5F2N3O2
- Molecular weight: 165.099
- Purity: 95%+
Ethyl azidodifluoroacetate introduces a fluorinated amide-triazole motif to drug candidates by amidation/click chemistry sequence.
Due to electron withdrawing effect of fluorine substituents, ethyl azidodifluoroacetate is very prone to facile hydrolysis and has acylating power comparable or better than that of NHS esters.
Therefore it is recommended to first leverage the acylating power, form amides from the corresponding amine partners and then subject the resulting amides to a subsequent click reaction.
